COVID-19
- HOPE Corona as an effective platform support
HOPE Corona supports the care of patients by linking all the tests and vaccinations of COVID-19 in a common digital care plan. The goal is to support the patient’s need for increased availability and service with high medical quality in the implementation of each individual activity.
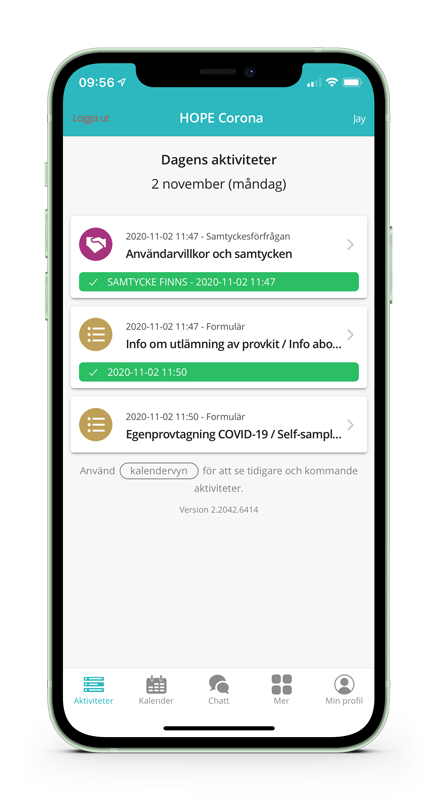
Digital care plan
The digital care plan enables the patient to easily become involved in their own self-care and as a contributor when testing and/or vaccinating for COVID-19. Care staff can quickly and efficiently monitor all planned and scheduled patient activities. The result is minimisation the recurring, manual administrative work for care professionals, allowing them to focus more on specialised activities.
Clinical decision support
Built-in clinical decision support is an additional tool based on automation rules generated by care staff activities. An alert is automatically sent to care staff via clinical decision support if patient medical activities and self-care do not follow the care plan set-up